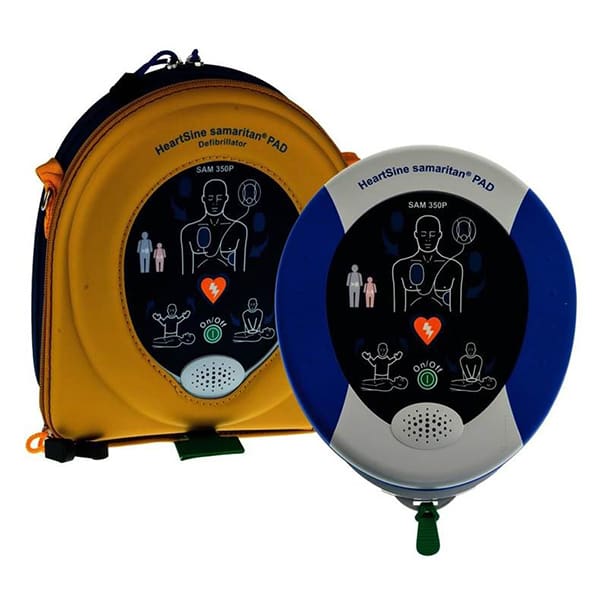

HeartSine samaritan® PAD 350P – Semi-Automatic Automated External Defibrillator
The HeartSine samaritan® PAD 350P features easy-to-understand visual and verbal prompts that guide a user through the resuscitation process. Utilizes biphasic technology. Suitable for adult and pediatric patients. Compact and lightweight. Comes with an 8-year warranty. Adult pads and carry case included. Pediatric pads sold separately.
Product Highlights
- Lighter (2.4 pounds) and smaller than other defibrillators.
- Resists shock and vibration and carries an IP56 rating, the industry’s highest rating against dust and water. It also carries an 8-year unit warranty.
- Uses proprietary electrode technology, advanced and stable firmware, and proprietary SCOPE™ Biphasic Technology to assess rhythm and recommend defibrillation if necessary.
- Features easy-to understand visual and voice prompts that guide a user through the process. Only two buttons, ON/OFF and SHOCK, are required.
- Pad-Pak™ cartridge combines battery and electrode pads, with one expiration date to monitor.
- Cartridge has a shelf life of four years from date of manufacture, offering significant savings over other defibrillators that require separate battery and pad units.
Specifications
Physical (With Pad-Pak™ Inserted)
Size: 8.0 inches by 7.25 inches by 1.9 inches (20 centimeters by 18.4 centimeters by 4.8 centimeters)
Weight: 2.4 pounds (1.1 kilogram) including Pad-Pak Battery
Defibrillator
Waveform: Self-compensating output pulse envelope biphasic waveform. Optimized biphasic escalating waveform compensates energy, slope and envelope for patient impedance.
Energy Selection
Adult: 1. Shock 150J 2. Shock 150J 3. Shock 200J
Pediatric: 1. Shock 50J 2. Shock 50J 3. Shock 50J
Charging Time
New Battery: Typically 150J in < 8 seconds, 200J in < 12 seconds
After 6 Discharges: Typically 150J in < 8 seconds, 200J in < 12 seconds
Patient Analysis System
Method: Evaluates patient’s ECG, signal quality, electrode contact integrity and patient impedance to determine if defibrillation is required.
Sensitivity/Specificity: Meets ISO 60601-2-4 and AAMI DF80:2003
Environmental
Operating/Standby Temperature: 0°C to 50°C (+32°F to +122°F)
Temporary Transportation Temperature: –10°C to 50°C (14°F to 122°F) for up to two days. Unit must be returned to standby/operating temperature for 24 hours before use.
Relative Humidity: 5% to 95% (non-condensing)
Water Resistance: IEC 60529/EN 60529 IP56
Altitude: 0 to 15,000 feet (0 – 4,575 meters)
Shock: MIL STD 810F Method 516.5, Procedure I (40G’s)
Vibration: MIL STD 810F Method 514.5+. Category 4 Truck Transportation – US Highways Category 7 Aircraft – Jet 737 & General Aviation (Exposure)
EMC: EN 60601-1-2, Second Edition: 2002
Radiated Emissions: EN55011:1999+A2:2001
Electrostatic Discharge RF Immunity: EN61000-4-3:2001 80MHZ-2.5GHZ (10V/m)
Magnetic Field Immunity: EN61000-4-8:2001 (3 A/m)
Aircraft: RTCA/DO-160F: 1997, Section 21 (Category M) RTCA DO-227
Falling Height: 1 meter
Event Documentation
Type: Internal Memory
Memory Capacity: 90 minutes of ECG (full disclosure) and event/incident recording
Playback Capabilities: Custom USB cable directly connected to PC and Saver™ EVO Windows-based data review software
Materials Used
PAD SAM350P: ABS, Santoprene. Printed circuit board with electronic components.
PAD Cartridge: Battery: lithium manganese dioxide
Housing: ABS – electrodes: hydrogel, silver, aluminium and polyester
Pad-Pak — Electrode and Battery Cartridge
Shelf Life: 4 years from manufacture date
Weight: 0.44 pounds (0.2 kilogram)
Size: 3.93 inches by 5.24 inches by .94 inches (10 centimeters by 13.3 centimeters by 2.4 centimeters)
Battery Type: Lithium Manganese Dioxide (LiMnO2)
Capacity: Meets ISO 60601-2-4 and AAMI DF80:2003 18V, 1.5 Amp Hrs
Electrodes: HeartSine samaritan® disposable defibrillation pads are supplied as standard with each device
Placement: Anterior-lateral (Adult); Anterior-posterior (Pediatric)
Active Gel Area: 100cm2
Cable Length: 3.5 feet (1 meter)
Always Up To Date
Auto-Replenishment Service
HealthFirst’s Auto-Replenishment Service uses smart automation to track your AED pads and batteries and automatically replenish them on time. Facilities benefit by lowering costs, saving time and gaining peace of mind.
OnTraq
OnTraq, HealthFirst’s online readiness application, reduces the risks, headaches and costs of emergency preparedness and regulatory compliance. Rely on OnTraq’s smart automation for dashboards, reports and task management.